Speed, Precision, & Results You Can Count On
Specializing in early-stage clinical research and high-quality bioanalytical data to move trials forward.

Our Team

Pete Boldingh
Vice President, Clinical Operations
Pete Boldingh, Pharm.D. has over 15 years in the clinical research industry. He has held past management positions with PRACS Institute, Ltd., Weber and Judd Pharmacies, and Vibra Hospital Pharmacy. Currently, Dr. Boldingh oversees clinical operations including study scheduling and management. His oversight includes pharmacy operations, volunteer recruitment, volunteer screening, training, SOP’s and strategic planning for clinical operations. He has served as a principle or sub-investigator on clinical projects for over 20 years. Dr. Boldingh is a registered pharmacist in Minnesota and North Dakota. He received his Doctor of Pharmacy degree from North Dakota State University, Fargo. He is a member of the Minnesota Pharmaceutical Association, North Dakota Pharmaceutical Association, and the Association of Clinical Research Professionals.

Melissa Burgard
Director, Clinical Research Services
Melissa Burgard, MSW, CCRC brings 30 years of Clinical Trial Operations Management and Cross Functional Team Leadership Experience. She started her research career in the therapeutic areas of Neurology and Psychiatry in Program Management, overseeing Federal Grant-Funded Research. In addition to her Psychiatry and CNS experience, Melissa has Trial Management and Therapeutic Expertise in the areas of Oncology & Immunotherapy, Infectious Disease & Vaccine, Ophthalmology, Pediatrics, Women’s Health, and Pulmonology. Melissa has provided Strategic and Executive Leadership for Clinical Trials conducted in Research Centers, Hospitals and Clinics across the United States. Her most recent appointment, prior to joining the AXIS Clinicals team in 2018, was as Director of Clinical Research Operations at Sanford Health, one of the largest healthcare systems in the Midwest. During her term at Sanford Health, she sat on an Advisory Board for Operational Activities for an Oncology Immunotherapy Network. Melissa maintains her Certificate as a Clinical Research Processional. She also serves as an elected Board Member for Moorhead Area Public Schools.

Amy Karels
Director, Quality Assurance
Amy Karels is the Quality Assurance Director at AXIS Clinicals LLC, bringing more than 26 years of experience in clinical research. She has spent 15 years in clinical operations, working across a diverse range of clinical studies and projects, which provides her with a deep, practical understanding of clinical trial conduct from both operational and quality perspectives. Throughout her career, Amy has been actively involved in sponsor qualification visits, regulatory authority inspections, and responses to regulatory queries, including interactions with the U.S. Food and Drug Administration (FDA), the World Health Organization (WHO), and regulatory authorities in Thailand and Malaysia. Her regulatory experience supports AXIS Clinicals’ commitment to inspection-readiness and global compliance. In her current role, Amy leads continuous improvement initiatives, with a strong focus on Corrective and Preventive Action (CAPA) management, risk mitigation, and quality systems enhancement. She is dedicated to ensuring compliance with ICH guidelines and FDA Good Clinical Practice (GCP) requirements, while promoting a culture of quality, accountability, and operational excellence.

Dr. Jim Carlson
Chief Scientific Advisor
James D. Carlson, Pharm.D provides over 35 years of experience in the clinical research industry. Dr. Carlson co-founded the PRACS Institute, LTD in Fargo in 1983 and assumed ownership and principal investigator status in 1990. He directed the growth of PRACS, which included adding e-DC, scientific affairs, regulatory affairs, bioanalytical sample analysis, data management, business development and dermatology studies. Dr. Carlson later served as President/CEO of Algorithme Pharma USA LLC in Fargo. Dr. Carlson received his Bachelors and Doctor of Pharmacy degrees from the Universities of Iowa and Michigan, respectively, and as a Clinical Pharmacy Residency from Buffalo General, NY. At AXIS Clinicals, Dr. Carlson aids in enhancing our clinical and bioanalytical services to support various studies while facilitating study designs and project execution of these programs. He is a member of the American Association of Pharmaceutical Scientists and the North Dakota Pharmaceutical Association.

John Pottier
Senior Vice President, Business Development
As a collaborative business development executive, John Pottier has over 30 years of leadership experience within the contact research industry. Identifying and implementing sales and marketing relationship strategies, John has built business models to serve the pharmaceutical markets’ clinical research requirements of NDA, Biosimilar, Biologics, regulatory filings and conduct pharmacology programs. Facilitating business development initiatives, John addressed clinical research markets within areas of cardiac safety, obesity, diabetes, dermatology, and neurology/psychology therapeutic areas. John earned a Bachelor of Science Business degree with a major in Marketing and is a member of American Association of Pharmaceutical Scientist, Bio International, Drug Information Association, Associates for Affordable Medicines.
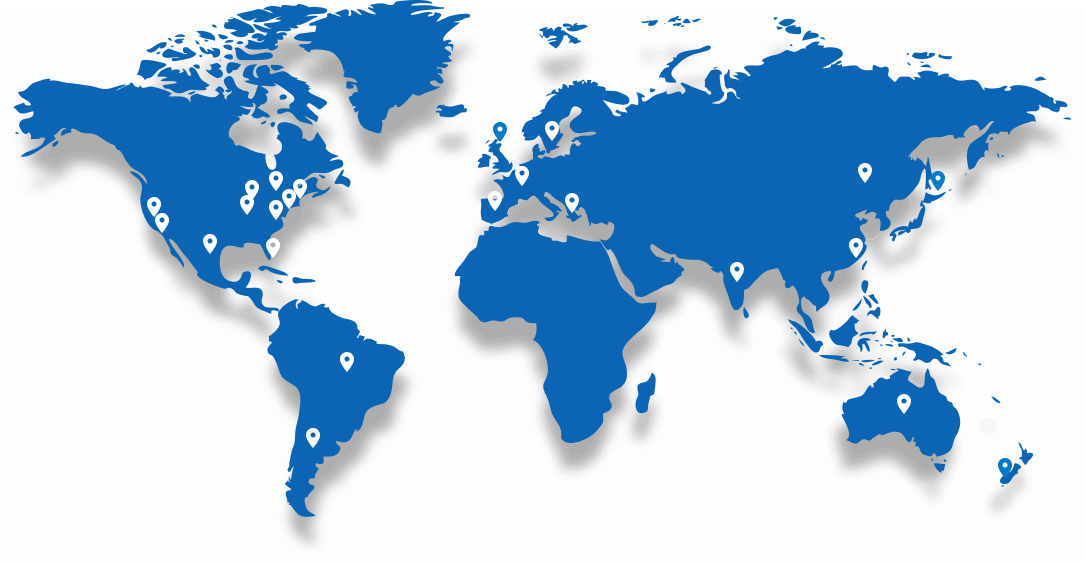
Serving Clients Around the World
Trusted by more than 100 Biotech and Pharmaceutical companies globally, AXIS supports clinical research with a reputation for speed, accuracy, and reliability.
With global reach and local expertise, we’ve earned the confidence of innovators across the US, Europe, Asia, and beyond.
Every Trial, Customized for You
No two studies are alike, which is why AXIS tailors each trial to meet your unique needs.
Flexible study design
Every protocol is carefully aligned to your therapeutic area, patient population, and development goals.
Adaptive recruitment strategies
Every protocol is carefully aligned to your therapeutic area, patient population, and development goals.
Seamless collaboration
Clear communication and dedicated teams keep your study moving forward on schedule and on budget.

Confident Results Backed by Decades of Experience
With three decades of clinical research experience, AXIS has built a strong foundation of knowledge, innovation, and trust.
Our teams bring together scientific rigor and operational excellence to accelerate development without sacrificing quality.
Take a look inside our clinical unit and see how our team delivers every study with precision and care.
Inside AXIS Clinicals
This short video highlights our facilities, our process, and the people behind the science. You’ll hear directly from staff who bring expertise and dedication to every trial—showing why AXIS is more than a site, it’s your trusted partner in moving research forward.
Trusted by Leading Innovators
See why top companies rely on Axis to accelerate trials deliver reliable results.












Experience by the Numbers
Behind every figure is a story of accelerated trials, confident decisions, and long-term collaboration.
Phase I / Early-Stage Studies

Expertly designed, executed, and monitored to deliver fast, reliable data.
Partner with AXIS for a trial that fits your goals.
Global Locations
Our strategic locations in the United States, India, and Mexico give you access to:

- Diverse patient populations
- Competitive timelines
- Cost-effective solutions
- Global regulatory expertise
AXIS India

Since 2004, AXIS India has delivered reliable, world-class clinical research solutions to sponsors across the globe.
With more than 3,500 studies completed for submissions in the US, Europe, Asia, Africa, and beyond, our team is trusted to provide accurate results—fast.
Backed by 500+ highly trained professionals, including project managers, bioanalytical scientists, and data specialists, AXIS India offers end-to-end support that streamlines every step of the clinical research process.
Our state-of-the-art facilities include:
- Two Clinical Pharmacology Units with 330+ beds for large-scale bioequivalence studies.
- 26 LC-MS/MS systems ensuring speed and precision in bioanalysis.
- 50+ clinical endpoint studies conducted across therapeutic areas such as oncology, CNS, gastroenterology, dermatology, ophthalmology, and respiratory.
With this depth of expertise and infrastructure, AXIS India helps sponsors accelerate development timelines while ensuring confidence in every result.
AXIS Mexico

Since 2011, AXIS Mexico has supported sponsors with 400+ studies conducted over the past decade, helping major pharmaceutical companies register products in Mexico and across Latin America.
Recognized by COFEPRIS and ISP Chile, we are a trusted partner for bioequivalence and clinical research in the region.
Our 70+ dedicated professionals bring deep expertise in clinical development and regulatory compliance, ensuring accurate results that meet global and regional standards.
- Two Clinical Pharmacology Units with 330+ beds for large-scale bioequivalence studies.
- Four advanced LC-MS/MS systems delivering reliable, high-quality bioanalytical data.
- The distinction of being the only CRO in Mexico approved for studies involving controlled substances.
With this unique positioning, AXIS Mexico provides sponsors with the speed, reliability, and compliance needed to move trials forward confidently in Latin America.
Meet the Team
Behind every successful study is a team that cares.
At Axis, our scientists and clinical experts are passionate about advancing research and helping clients achieve their milestones faster, with confidence.

Have questions or need expert support? Our team is here to help—reach out and let’s move forward together.