Speed, Precision, & Results You Can Count On
Specializing in early-stage clinical research and high-quality bioanalytical data to move trials forward.

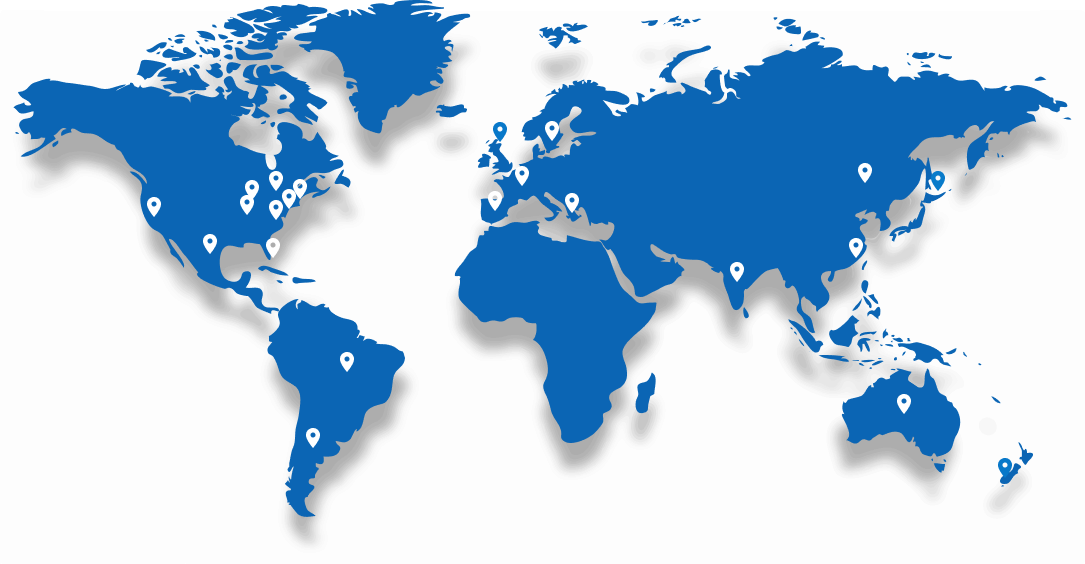
Serving Clients Around the World
Trusted by more than 100 Biotech and Pharmaceutical companies globally, AXIS supports clinical research with a reputation for speed, accuracy, and reliability.
With global reach and local expertise, we’ve earned the confidence of innovators across the US, Europe, Asia, and beyond.
Every Trial, Customized for You
No two studies are alike, which is why AXIS tailors each trial to meet your unique needs.
Flexible study design
Every protocol is carefully aligned to your therapeutic area, patient population, and development goals.
Adaptive recruitment strategies
Every protocol is carefully aligned to your therapeutic area, patient population, and development goals.
Seamless collaboration
Clear communication and dedicated teams keep your study moving forward on schedule and on budget.

Confident Results Backed by Decades of Experience
With three decades of clinical research experience, AXIS has built a strong foundation of knowledge, innovation, and trust.
Our teams bring together scientific rigor and operational excellence to accelerate development without sacrificing quality.
Take a look inside our clinical unit and see how our team delivers every study with precision and care.
Inside AXIS Clinicals
This short video highlights our facilities, our process, and the people behind the science. You’ll hear directly from staff who bring expertise and dedication to every trial—showing why AXIS is more than a site, it’s your trusted partner in moving research forward.
Trusted by Leading Innovators
See why top companies rely on Axis to accelerate trials deliver reliable results.












Experience by the Numbers
Behind every figure is a story of accelerated trials, confident decisions, and long-term collaboration.
Phase I / Early-Stage Studies

Expertly designed, executed, and monitored to deliver fast, reliable data.
Partner with AXIS for a trial that fits your goals.
Global Locations
Our strategic locations in the United States, India, and Mexico give you access to:

- Diverse patient populations
- Competitive timelines
- Cost-effective solutions
- Global regulatory expertise
AXIS India

Since 2004, AXIS India has delivered reliable, world-class clinical research solutions to sponsors across the globe.
With more than 3,500 studies completed for submissions in the US, Europe, Asia, Africa, and beyond, our team is trusted to provide accurate results—fast.
Backed by 500+ highly trained professionals, including project managers, bioanalytical scientists, and data specialists, AXIS India offers end-to-end support that streamlines every step of the clinical research process.
Our state-of-the-art facilities include:
- Two Clinical Pharmacology Units with 330+ beds for large-scale bioequivalence studies.
- 26 LC-MS/MS systems ensuring speed and precision in bioanalysis.
- 50+ clinical endpoint studies conducted across therapeutic areas such as oncology, CNS, gastroenterology, dermatology, ophthalmology, and respiratory.
With this depth of expertise and infrastructure, AXIS India helps sponsors accelerate development timelines while ensuring confidence in every result.
AXIS India

Since 2011, AXIS Mexico has supported sponsors with 400+ studies conducted over the past decade, helping major pharmaceutical companies register products in Mexico and across Latin America.
Recognized by COFEPRIS and ISP Chile, we are a trusted partner for bioequivalence and clinical research in the region.
Our 70+ dedicated professionals bring deep expertise in clinical development and regulatory compliance, ensuring accurate results that meet global and regional standards.
- Two Clinical Pharmacology Units with 330+ beds for large-scale bioequivalence studies.
- Four advanced LC-MS/MS systems delivering reliable, high-quality bioanalytical data.
- The distinction of being the only CRO in Mexico approved for studies involving controlled substances.
With this unique positioning, AXIS Mexico provides sponsors with the speed, reliability, and compliance needed to move trials forward confidently in Latin America.
Meet the Team
Behind every successful study is a team that cares.
At Axis, our scientists and clinical experts are passionate about advancing research and helping clients achieve their milestones faster, with confidence.

Have questions or need expert support? Our team is here to help—reach out and let’s move forward together.