PHARMACOLOGY STUDIES
Accelerating Clinical Research Without Compromise
Your CRO in clinical research dedicated to fast recruitment, clean data, and confident milestones.

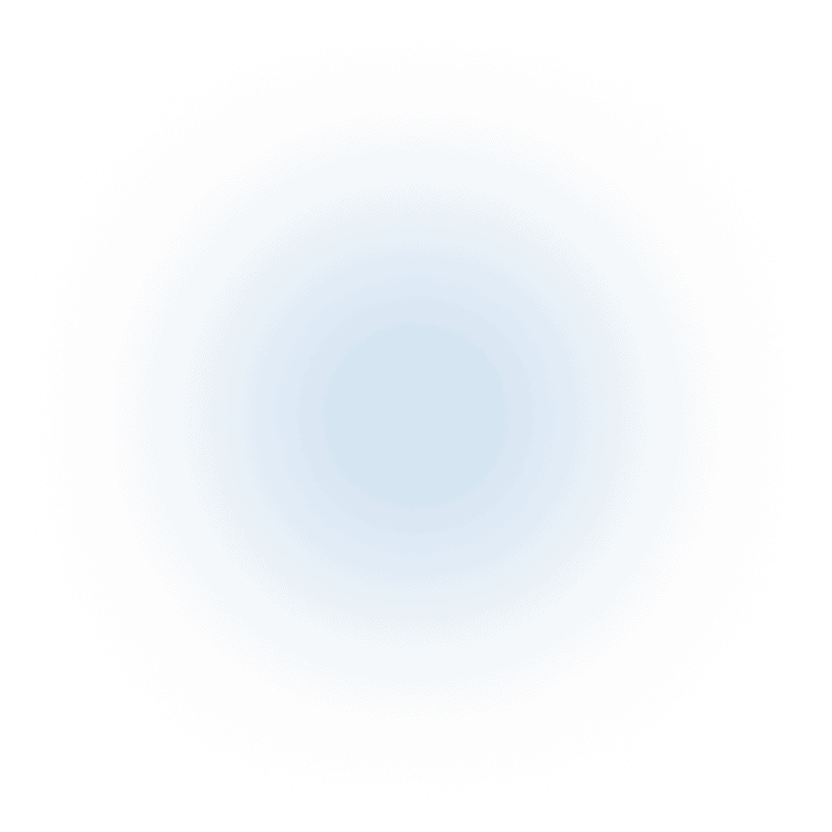
You Need a Clinical Pharmacology CRO Built to Deliver Speed Without Losing Data Quality
The early stages of drug development demand speed and precision.
AXIS becomes part of your team—bringing fast, high-quality pharmacology services that keep your program on track.
Get Every Early-Stage Pharmacology Study You Need to
Advance Your Drug Development
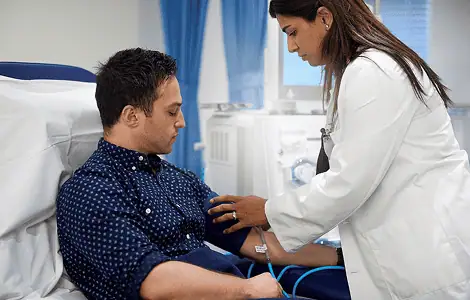
Dedicated clinical pharmacology unit equipped for efficient Phase I and early-stage trials.


- First-in-Human
- Food Effect
- Pharmacodynamics
- hADME
- Drug-Drug Interactions
- Bioavailability / Bioequivalence
- Linearity and Dose Proportionality
- Single and Multiple Ascending Dose
- High Precision TQT, Cardiac safety
- Dermal Open Flow Microperfusion
Keeps Your Trial Moving—Fast
With recruitment strategies designed to get you to first-patient-in quickly and keep enrollment on track, you can hit critical milestones faster and maintain momentum throughout your trial.
Our clinical pharmacology unit services combine the strength of an expansive volunteer database with the advantage of our strategic location near major universities and diverse communities.
This proactive approach means fewer delays, stronger retention, and the confidence that your enrollment targets will be met on time, helping your study move forward without disruption.
Expansive Volunteer Database
Access a large, engaged database of healthy volunteers, ensuring enrollment targets are met without delays.
Strategic Location
Our clinical research center, near major universities and diverse populations, provides access to a broad pool of eligible participants.
Community Involvement
Strong ties with local communities help us build trust, increase participation, and sustain recruitment momentum across studies.
Quick Start-up
With optimized processes, you’ll have your first patients enrolled within weeks, not months.
Specialized Expertise in Clinical Pharmacology
When you’re advancing a new therapy, you need a partner who understands the complexity of early-stage development and can deliver results with precision.
From NCEs and NBEs to biosimilars, 505(b)(2) products, and repurposed compounds, our team has successfully conducted pharmacology studies across a wide range of therapies.
With board-certified dermatologists and highly trained staff, we execute studies for topical and transdermal products with accuracy and efficiency.
Our portfolio includes every major clinical pharmacology study design: SAD/MAD, DDI, dose proportionality, food-effect (FE), hADME, and TQT.
Axis is the only CRO in the United States licensed to perform dermal Open Flow Microperfusion (dOFM) studies, giving sponsors exclusive access to this cutting-edge methodology.


Axis is the only CRO in the United States licensed to perform dermal Open Flow Microperfusion (dOFM) studies.
hADME Pharmacology Studies
Better Answers, Sooner

Clinical pharmacology unit services backed by experienced staff and proven expertise.
hADME studies are critical to predicting how a drug behaves in the body—and we make sure you’re ahead of the curve. At AXIS, our hADME services help you:
- Design smarter formulations based on how your drug is absorbed, distributed, metabolized, and excreted.
- Enhance safety and dosing precision, reducing the risk of setbacks and boosting confidence in your next stage.
- Streamline clinical trial strategy, by using hADME data to shape efficient, patient-focused study designs.
Our expert team—supported by dedicated clinical trial units, monitoring protocols, and hot labs for sample analysis—acts as an extension of your organization from Day 1.
Faster insights, tighter timelines, and more clarity on your path to development.
dOFM Pharmacology Studies
Revolutionizing drug development with precision and speed
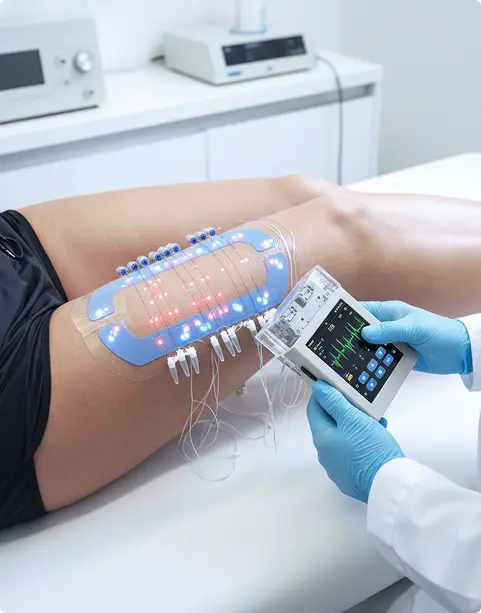
Clinical pharmacology services that provide the insights you need to make confident decisions, quickly.
- Smaller, smarter studies to capture meaningful, statistically relevant human data with minimal subjects, saving both time and cost compared to traditional endpoint approaches.
- Deeper insight into drug behavior by continuously sampling dermal interstitial fluid right at your drug’s site of action, dOFM reveals how your formulation truly performs, helping you make informed development decisions earlier.
Reliable Cardiovascular Monitoring
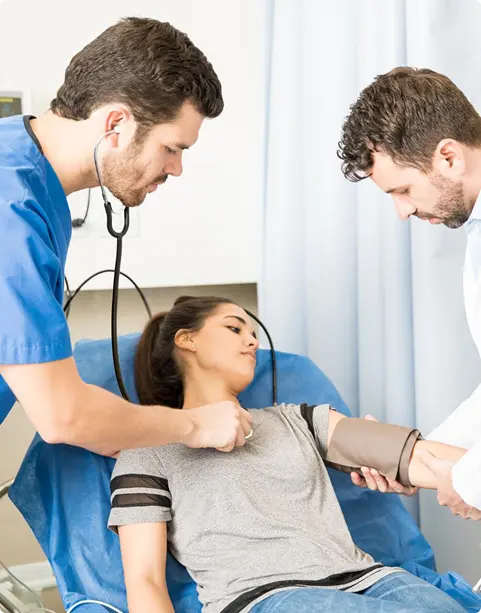
Precise ECG clinical trial data to ensure reliable cardiac safety results.
Thorough TQT studies are essential for evaluating the cardiac safety of new therapies. At Axis, we design and conduct TQT studies with precision, leveraging advanced ECG clinical trial capabilities to detect even subtle effects on cardiac repolarization.
Why this matters for your program:
- High-quality cardiac safety datayou can trust for regulatory submissions
- Seamless ECG integration for precise, real-time monitoring
- Proven expertise in designing and executing TQT studies efficiently
Technology + Expertise for Real-Time Data & Reliable Results
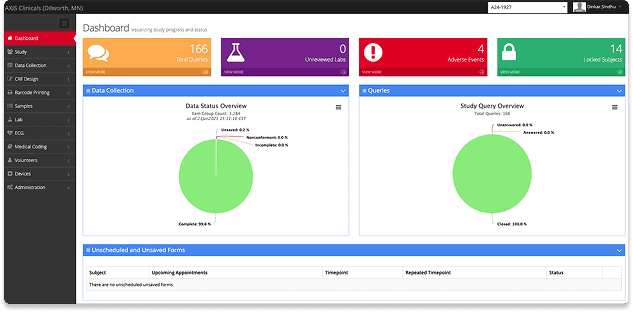
When your trial depends on accurate, real-time data, you need a system that ensures quality from start to finish. At AXIS, our integrated technology and expert processes keep your study moving forward with clarity and confidence.
Accurate, Real-Time Data
Every study is captured electronically in ClinSpark®, the eSource/EDC platform. Seamless integration with medical devices means real-time collection and fewer errors.
Safety You Can Trust
Bi-directional connectivity with our clinical safety lab ensures continuous monitoring and compliance with CDISC standards, so you always have a clear picture of patient safety.
Consistency from the Start
Our pharmacists design robust drug preparation, compounding, and dosing protocols, then train staff to ensure dosing is consistent across every subject and study.
Customized for Every Project
Each study begins with a structured meeting to align communication and workflow. From planning to execution, our priority is delivering precision, regulatory approval, and stakeholder confidence.
High-Quality, Consistent Data with eSource and EDC System
State-of-the-art clinical pharmacology research unit supporting innovative early-stage studies.
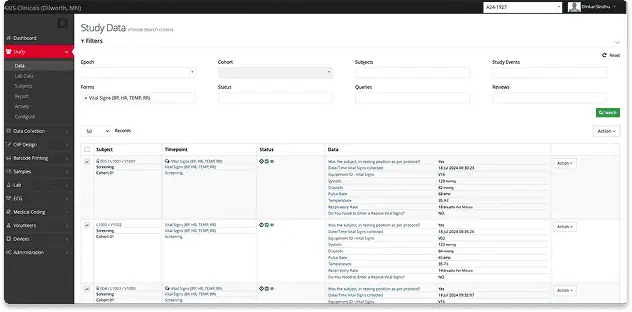
Electronic Data Capture
All study data is collected electronically in ClinSpark®, the industry-leading eSource/EDC platform built for Phase I trials.
Seamless Device Integration
Direct connections with ECGs, vitals monitors, and scales allow for real-time data capture while eliminating transcription errors.
Continuous Safety Oversight
Bi-directional connectivity with our clinical safety lab—using CDISC and HL7 standards—ensures ongoing, real-time safety monitoring throughout your study.
Keep your trial on schedule with a Clinical Pharmacology CRO built for speed and precision.
Full-Scope Clinical Research CRO Services
We handle everything from the big picture to the smallest detail.

- Clinical Development and Strategic Consulting
- Clinical Trial Management
- Project Management
- Support Services: Labs, IP Management
- Clinical Monitoring
- Medical Monitoring
- Data Management
- Biostats and Medical Writing
- QA & Regulatory Submissions
Starting a study shouldn’t slow you down. At AXIS, we make the process clear, efficient, and designed for speed, so you can focus on your science, not bottlenecks.
Get Your Pharmacology Study Moving Fast
Starting a study shouldn’t slow you down. At AXIS, we make the process clear, efficient, and designed for speed, so you can focus on your science, not bottlenecks.

Schedule a
Consultation
Share your objectives and study
requirements with our experts.

Get a Custom Proposal
& Trial Timeline
We’ll outline a clear plan built
around your study’s needs.

Launch Quickly and
Stay on Track
Progress toward your next
milestone with high-quality data.
Don’t Let Missed Milestones Derail Your Phase 1 Trial
Partner with a Clinical Pharmacology CRO that accelerates timelines and ensures reliable results.

Free Guide:
5 Ways to Keep Your Phase I Trial on Schedule
A timely trial protects your milestones, budget, and momentum.
- Download our free guide and learn how to:
- Start faster without sacrificing quality
- Avoid common operational bottlenecks
- Keep your trial moving toward the next phase
Have questions or need expert support? Our team is here to help—reach out and let’s move forward together.