DERMATOLOGY STUDIES
Built for Speed and Accuracy
Reliable dermatology clinical trials delivering submission-ready data without delays.
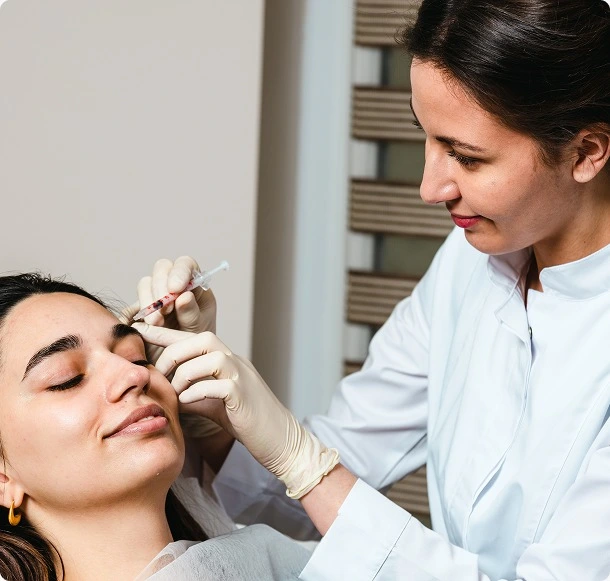
Strong Recruitment and High-Quality Data Keep Your Dermatology Studies on Track
Many dermatology clinical studies stall because enrollment, assessments, or data capture can’t keep pace with the trial’s demands.
At Axis, we’ve built the systems and expertise to prevent delays before they happen.
Early-Stage Dermatology Clinical Studies to
Keeps Your Trial Moving—Fast
- Vasoconstrictor Studies With more than 50 completed studies for topical corticosteroid products, our team brings unmatched experience in evaluating potency and performance.
- Adhesion Studies Our trained staff are experts in adhesion scoring, making us uniquely qualified to perform accurate, regulatory-compliant adhesion studies for transdermal delivery systems.
- Dermal Open Flow Microperfusion (dOFM) Unlike traditional methods, dOFM provides continuous, time-resolved PK/PD data at the drug’s site of action—delivering deeper insights into dermal drug behavior.
- Irritation & Sensitization Studies Our outpatient unit is built to seamlessly conduct large-scale trials, enrolling up to 300 volunteers to meet FDA guidance and deliver timely results.
Rapid Recruitment Keeps Your Dermatology Clinical Trial on Track
Quick enrollment is critical to maintaining momentum in dermatology clinical trials.
At Axis, our proactive recruitment strategies are designed to accelerate enrollment and keep it steady—so you can reach milestones faster without disruption.
With a large volunteer database, a strategic location near major universities, and strong community partnerships, we ensure your enrollment goals are met on time and with confidence.
- Expansive Volunteer Database Access a broad, engaged pool of healthy volunteers to meet enrollment targets without delays.
- Strategic Location Our site’s proximity to universities and diverse communities provides a steady stream of eligible participants.
- Community Involvement Long-standing community ties build trust and drive consistent participation across studies..
- Quick Start-up Optimized processes mean your first patients are enrolled within weeks, not months.


With Axis, your dermatology clinical studies meet FDA requirements faster with rapid recruitment of 300 volunteers.
dOFM Dermatology Studies

Trusted Partner for Global Dermatology Clinical Studies search With Speed and Precision
Transforming Dermatology ReYou need more than a CRO—you need a partner who works as an extension of your team, bringing expertise and precision to every dermatology clinical trial.
- Specialized Facilities A 6,000 sq ft dermatology unit designed for both outpatient and inpatient studies.
- Expert Staff Dermatology-trained teams skilled in scoring and evaluation methods.
- Faster Timelines Efficient recruitment, streamlined processes, and rapid study start-up keep your trial on track.
- High-Quality Data Submission-ready results collected with precision and consistency to support confident regulatory approval.
Reliable Systems & Technology for Faster, Cleaner Studies
Behind every successful dermatology clinical trial is the right combination of technology and expertise. Our seamless systems and experienced staff work together to deliver high-quality data you can trust—quickly and consistently—every subject, every study, every time.
Accurate, Real-Time Data
Every study is captured electronically in ClinSpark®, the eSource/EDC platform. Seamless integration with medical devices means real-time collection and fewer errors.
Safety You Can Trust
Our clinical safety lab ensures continuous monitoring and compliance with CDISC standards with Bi-directional connectivity, so you always have a clear view of participant safety.
Consistency from the Start
Our pharmacists design and validate robust preparation, compounding, and dosing protocols, then train staff to ensure dosing is consistent across every subject, every time.
Customized for Every Project
Each dermatology clinical study begins with a structured kick-off to align communication and workflow. From protocol design to final submission, our focus is delivering speed, precision, and high-quality results.
High-Quality Data You Can Trust With Integrated eSource + EDC Systems
Electronic Data Capture
All dermatology study data is collected electronically in ClinSpark®, the industry-leading eSource/EDC platform built for Phase I trials.
Seamless Device Integration
Direct integration with ECGs, vitals monitors, and scales ensures real-time data capture, eliminating manual transcription errors and increasing accuracy.
Continuous Safety Oversight
Bi-directional connectivity with our clinical safety lab—using CDISC and HL7 standards—provides ongoing safety monitoring throughout every stage of your study.
Advance your dermatology clinical trials with a CRO that delivers speed, accuracy, and submission-ready data.
Complete Clinical Research CRO Services & Partner
We streamline complex processes and manage every detail to keep your study on track.

Expertise You Can Count On for Dermatology Clinical Studies
- Clinical Development and Strategic Consulting
- Clinical Trial Management
- Project Management
- Support Services: Labs, IP Management
- Clinical Monitoring
- Medical Monitoring
- Data Management
- Biostats and Medical Writing
- QA & Regulatory Submissions
The Fast Track to Dermatology Clinical Studies
Dermatology clinical trials should accelerate your progress, not create delays. At Axis, we simplify every step with clear processes and rapid execution, so your studies move forward smoothly and you stay focused on advancing science.

Schedule a
Consultation
Share your objectives and study
requirements with our experts.

Get a Custom Proposal
& Trial Timeline
We’ll outline a clear plan built
around your study’s needs.

Launch Quickly and
Stay on Track
Progress toward your next
milestone with high-quality data.
Don't Let Delays and Errors Put Your Trail at Risk
Partner with a Clinical Dermatology CRO that accelerates timelines and ensures accurate results.

Free Guide:
5 Ways to Keep Your Phase I Trial on Schedule
A timely trial protects your milestones, budget, and momentum.
- Download our free guide and learn how to:
- Start faster without sacrificing quality
- Avoid common operational bottlenecks
- Keep your trial moving toward the next phase
Have questions or need expert support? Our team is here to help—reach out and let’s move forward together.